Enzyme Activity
Enzymes are part of the group of chemicals called proteins. They are any class of nitrogenous organic compounds which have large molecules composed of one or more long chains of amino acids. Proteins, along with carbohydrates and fats, are the chemicals that make up most of the diet of animals. Proteins are made of amino acids, of which 20 different kinds exist in human cells.
Essentially there are two groups of protein: fibrous and globular.
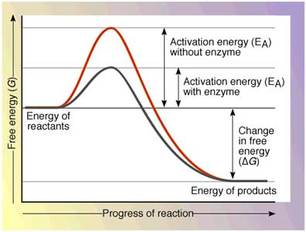
These essential functions make proteins very important. Proteins are coded for by DNA, with one gene of DNA coding for one specific protein; they are synthesised by ribosomes. Enzymes act as biological catalysts, controlling the speed of chemical reactions in all organisms. Enzymes increase the rate of chemical reactions- without them, metabolism would take place too slowly for life to exist. Chemical reactions only occur when the enzyme and substrate collide in the correct orientation with the minimum activation energy. Energy enables a lower activation energy requirement for the reaction by providing an alternative pathway for the reaction.
Metabolism (metabolic reactions) refers to all the chemical reactions occurring in a cell (e.g DNA replication, protein synthesis, respiration, photosynthesis). All the metabolic reactions of a cell are controlled by enzymes.
Essentially there are two groups of protein: fibrous and globular.
- Fibrous- these are long and stringy and form many of the structural elements of the body (e.g collagen in muscles and tendons; elastic in the connective tissue of the skin; keratin in hair, horns, nails)
- Globular- these are much-folded into a ball shape, and perform many of the regulatory processes of the body (e.g hormones such as insulin, transporters such as haemoglobin, enzymes such as sucrase; antibodies, antitoxins).
These essential functions make proteins very important. Proteins are coded for by DNA, with one gene of DNA coding for one specific protein; they are synthesised by ribosomes. Enzymes act as biological catalysts, controlling the speed of chemical reactions in all organisms. Enzymes increase the rate of chemical reactions- without them, metabolism would take place too slowly for life to exist. Chemical reactions only occur when the enzyme and substrate collide in the correct orientation with the minimum activation energy. Energy enables a lower activation energy requirement for the reaction by providing an alternative pathway for the reaction.
Metabolism (metabolic reactions) refers to all the chemical reactions occurring in a cell (e.g DNA replication, protein synthesis, respiration, photosynthesis). All the metabolic reactions of a cell are controlled by enzymes.
- The reactions that synthesise (build) large molecules from smaller ones (e.g proteins from amino acids in protein synthesis) are anabolism (anabolic reactions).
- The reactions that break down large molecules into smaller ones (e.g carbohydrates into glucose in digestion) are catabolism (catabolic reactions).
Enzymes are specific, i.e one enzyme catalyses (increases the rate of a chemical reaction without itself undergoing any permanent chemical change) only one type of reaction. An enzyme is usually named after the substance it catalyses, by having the suffix "-ase" added, for example:
The reason that enzymes are specific relates to their shape. Each enzyme has a particular shape, determined by the sequence of amino acids that it is made of. The amino acids in a protein may be cross-linked by hydrogen bonds (in some cases sulfur bonds), and this is what gives the protein its distinctive shape.
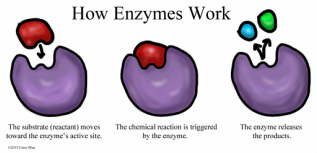
The shape of the enzyme in an area known as its active site corresponds to that of the substance(s) it catalyses- binding to the protein or other substance during a reaction in the lock-and-key model of enzyme action. In the way that a key fits into a particular lock, so only one specific substrate (molecule which an enzyme acts on) fits into the active site of the enzyme.
Another way of enzymes is when before binding, the substrate molecules and enzymes do not exactly fit each other. As the enzyme and substrate fit together (forming the enzyme-substrate complex), chemical bonds form or are broken. The enzyme's active site changes it's shape slightly (through the weak hydrogen bonds that hold the shape) when combined with the substrate to fit (called an induced fit). This distorts the substrate molecule(s), making reactions more likely (the more accepted model of enzyme action). The final product afterwards is released and the enzyme returns to its previous shape. Different molecules cannot induced a fit with the enzyme.
Enzymes are not consumed or broken down in reactions, so one enzyme molecule can catalyse the same reaction many times and do do at a vary fast rate (e.g peroxidase in liver cells can catalyse the breakdown of several million hydrogen peroxide molecules into water and oxygen every minute. Hydrogen peroxide is a toxic substance that harms the liver/body unless it is broken down rapidly into water and oxygen which the body can use). This saves the cell resources and energy from not having to constantly synthesise new enzymes. The enzyme provided the template or "work bench" for substrate molecules to react on.
Because their shape is maintained by weak hydrogen bonds, enzymes as susceptible to denaturing at high temperatures. As the hydrogen bonds are broken and the enzyme loses its shape, it can no longer catalyse reactions.
- Lipase catalyses the catabolism of lipids into fatty acids and glycerol.
- Maltase catalyses the catabolism of the sugar maltose into two glucose molecules.
- Peroxidase catalyses the breakdown of hydrogen peroxide, H202, into water and oxygen.
- Ligase catalyses the bonding of Okazaki fragments in DNA replication.
- Amylase catalyses the digestion (breakdown) of starch.
The reason that enzymes are specific relates to their shape. Each enzyme has a particular shape, determined by the sequence of amino acids that it is made of. The amino acids in a protein may be cross-linked by hydrogen bonds (in some cases sulfur bonds), and this is what gives the protein its distinctive shape.
The shape of the enzyme in an area known as its active site corresponds to that of the substance(s) it catalyses- binding to the protein or other substance during a reaction in the lock-and-key model of enzyme action. In the way that a key fits into a particular lock, so only one specific substrate (molecule which an enzyme acts on) fits into the active site of the enzyme.
Another way of enzymes is when before binding, the substrate molecules and enzymes do not exactly fit each other. As the enzyme and substrate fit together (forming the enzyme-substrate complex), chemical bonds form or are broken. The enzyme's active site changes it's shape slightly (through the weak hydrogen bonds that hold the shape) when combined with the substrate to fit (called an induced fit). This distorts the substrate molecule(s), making reactions more likely (the more accepted model of enzyme action). The final product afterwards is released and the enzyme returns to its previous shape. Different molecules cannot induced a fit with the enzyme.
Enzymes are not consumed or broken down in reactions, so one enzyme molecule can catalyse the same reaction many times and do do at a vary fast rate (e.g peroxidase in liver cells can catalyse the breakdown of several million hydrogen peroxide molecules into water and oxygen every minute. Hydrogen peroxide is a toxic substance that harms the liver/body unless it is broken down rapidly into water and oxygen which the body can use). This saves the cell resources and energy from not having to constantly synthesise new enzymes. The enzyme provided the template or "work bench" for substrate molecules to react on.
Because their shape is maintained by weak hydrogen bonds, enzymes as susceptible to denaturing at high temperatures. As the hydrogen bonds are broken and the enzyme loses its shape, it can no longer catalyse reactions.
Factors that Affect Enzyme Activity
Temperature
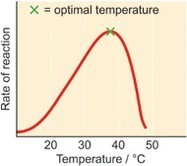
The warmer the temperature (usually up to about 40-45 degrees Celsius), the faster the enzymes catalyse a reaction. This is because increasing the temperature increases the speed at which the reacting particles move, so they collide more often. It is necessary for particles to collide for them to react. However, above 40-45 degrees Celsius, enzymes are usually denatured and can no longer catalyse the reaction, because the hydrogen bonds are broken, so the enzyme permanently loses its shape.
The temperature at which the reaction rate is fastest is the optimum temperature.
In homeotherms ("warm-blooded" animals- birds and mammals), the optimum temperature is their core body temperature (37 degrees Celsius in humans).
In polikilotherms ("cold-blooded" animals/organisms), the optimum temperature will depend on their habitat; it may be low if the organism lives in cool habitats (e.g some fish in the Antarctic seas have an optimum temperature approaching 0 degrees Celsius), and high in hot habitats (e.g bacteria that inhabit geothermal hot pools have an optimum temperature approaching 100 degrees Celcius).
The warmer the temperature (usually up to about 40-45 degrees Celsius), the faster the enzymes catalyse a reaction. This is because increasing the temperature increases the speed at which the reacting particles move, so they collide more often. It is necessary for particles to collide for them to react. However, above 40-45 degrees Celsius, enzymes are usually denatured and can no longer catalyse the reaction, because the hydrogen bonds are broken, so the enzyme permanently loses its shape.
The temperature at which the reaction rate is fastest is the optimum temperature.
In homeotherms ("warm-blooded" animals- birds and mammals), the optimum temperature is their core body temperature (37 degrees Celsius in humans).
In polikilotherms ("cold-blooded" animals/organisms), the optimum temperature will depend on their habitat; it may be low if the organism lives in cool habitats (e.g some fish in the Antarctic seas have an optimum temperature approaching 0 degrees Celsius), and high in hot habitats (e.g bacteria that inhabit geothermal hot pools have an optimum temperature approaching 100 degrees Celcius).
pH
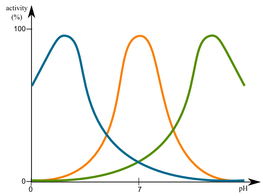
Most enzymes work with cells, so their optimum pH is approximately 7 (i.e close to neutral); exceptions are the digestive enzymes (e.g pespsin).
The specific shape of an enzyme is held together by the attraction between positive and negative charges on different parts of the molecule. The pH of the surrounding environment can affect the charges on the enzyme molecule. This can cause the protein chain to unfold or uncoil (denature). Even a slight change in an enzymes shape can prevent it from working properly.
Outside the optimum, enzyme activity will decrease. The enzyme will often change shape but does not normally denature. If the pH is outside the range for an enzyme, the enzyme denatures, and can no longer act as a catalyst.
Pepsin breaks down protein in the stomach and works well in acidic conditions. The stomach contents have a low pH because the presence of dilute hydrochloric acid.
The optimum pH for amylase (an enzyme in saliva, which breaks down starch) is about 7. Saliva has a neutral pH.
Pancreatic lipase breaks down lipids in the small intestine, where conditions are slightly basic (pH is greater than 7), because of the presence of bicarbonate ions.
Most enzymes work with cells, so their optimum pH is approximately 7 (i.e close to neutral); exceptions are the digestive enzymes (e.g pespsin).
The specific shape of an enzyme is held together by the attraction between positive and negative charges on different parts of the molecule. The pH of the surrounding environment can affect the charges on the enzyme molecule. This can cause the protein chain to unfold or uncoil (denature). Even a slight change in an enzymes shape can prevent it from working properly.
Outside the optimum, enzyme activity will decrease. The enzyme will often change shape but does not normally denature. If the pH is outside the range for an enzyme, the enzyme denatures, and can no longer act as a catalyst.
Pepsin breaks down protein in the stomach and works well in acidic conditions. The stomach contents have a low pH because the presence of dilute hydrochloric acid.
The optimum pH for amylase (an enzyme in saliva, which breaks down starch) is about 7. Saliva has a neutral pH.
Pancreatic lipase breaks down lipids in the small intestine, where conditions are slightly basic (pH is greater than 7), because of the presence of bicarbonate ions.
Substrate Concentration
The rate if enzyme activity will increase as the concentration of the substance or enzymes increases. This occurs up until a saturation point occurs (i.e no free enzymes/active sites). The more substrate molecules that are available, the more frequently they collide with the active site of the enzyme molecules. The higher the concentration of enzymes the more active sites there are to bind with the substrate. Both of these increase the enzyme activity causing an increase in rate of reaction. Saturation occur when the substrate occupies all the available active sites on all the available enzymes.
The rate if enzyme activity will increase as the concentration of the substance or enzymes increases. This occurs up until a saturation point occurs (i.e no free enzymes/active sites). The more substrate molecules that are available, the more frequently they collide with the active site of the enzyme molecules. The higher the concentration of enzymes the more active sites there are to bind with the substrate. Both of these increase the enzyme activity causing an increase in rate of reaction. Saturation occur when the substrate occupies all the available active sites on all the available enzymes.
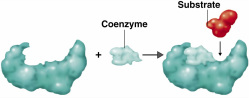
Co-factors
Many enzymes need another molecule to assist in catalysis . They help enzymes to function correctly. Co-factors work by binding to the active site of the enzyme and participating in catalysis but are not considered substrates of the reaction as they can be used again. These co-factors may be (small) inorganic ions such as cobalt, Co, selenium, Se, (can be ions such as magnesium, copper, iron) or (large) organic molecules such as some vitamins (FAD, coenzyme q10, haem). These organic co-factors are called co-enzymes.
Co-enzymes are necessary when only weak bonds form between the enzyme and substrate; the co-enzyme acts as a bridge, locking the enzyme and substrate more tightly together.
Many enzymes need another molecule to assist in catalysis . They help enzymes to function correctly. Co-factors work by binding to the active site of the enzyme and participating in catalysis but are not considered substrates of the reaction as they can be used again. These co-factors may be (small) inorganic ions such as cobalt, Co, selenium, Se, (can be ions such as magnesium, copper, iron) or (large) organic molecules such as some vitamins (FAD, coenzyme q10, haem). These organic co-factors are called co-enzymes.
- The carrier molecule NAD (required in respiration) is a co-enzyme derived from nicotinic acid, a member of the vitamin B complex.
- Hydrolases, which control a transcription factor that "turns on" specific genes, need vitamin C to be achieved.
Co-enzymes are necessary when only weak bonds form between the enzyme and substrate; the co-enzyme acts as a bridge, locking the enzyme and substrate more tightly together.
Inhibitors
Inhibitors are substances that prevent enzymes catalysing reactions, and so are poisons. they disrupt the active site so it is no-longer available for use by substrate. The enzyme becomes non-functional.
An inhibitor can act in the following ways.
Inhibitors are substances that prevent enzymes catalysing reactions, and so are poisons. they disrupt the active site so it is no-longer available for use by substrate. The enzyme becomes non-functional.
- Heavy metal ions (e.g lead, Pb, and mercury, Hg) prevent enzymes in cells of the nervous system functioning.
- Cyanide prevents the action of an enzyme in the electron transfer chain of respiration (causing death, as respiration ceases).
- Hemoglobin (a protein found in red blood cells that binds oxygen gas and transports it to our cells) is competitively inhibited in a similar manner by carbon monoxide gas.
An inhibitor can act in the following ways.
- Taking over the active site of the enzyme, so stopping the substrate from binding to the active site- e.g the antibiotic penicillin inhibits an enzyme that bacteria use to make their cell walls.
- Bonding to another part of the enzyme and altering the shape of the active site so that it can no longer bind to the substrate- this is usually temporary, and can be a way in which the cell can control a metabolic pathway.
